超说明书用药医疗损害责任实证研究
——以司法案例为研究基础

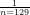
打开文本图片集
[中图分类号]R-052 [文献标志码]A [文章编号]1001-8565(2025)11-1431-10
DOI:10.12026/j.issn.1001-8565.2025.11.08
Abstract: The use of of-label drugs,which accompanies the approval system for new drugs entering the market,iswidespread in clinical practice in China,and the resulting medical damage liability is also controversial in theory and practice.Based on 129 judicial cases from 2O14 to 2023,this paper conducted a quantitative analysis of medical damage liability caused by of-label drug use.Through sorting out,it was found thatthecases ofoff-label drug use presented obvious characteristics in types and regional distribution,and their damage consequences and appeal rates were far higher than those of general medical damage cases,which need to attract more attention.Based on the liability reasons,this paper further categorized the cases into three types : no liability,single liability,and multiple liabilities.The number and liability ratioofof-label drug useunder different iability types were analyzed,and the key factors affcting the establishment of medical damage liability for off-label drug use were sorted out and analyzed in depth.These key factors included the existence of offlabel drug use and a clear causal relationship between off-label drug useand medical damage,inadequate informed consent,and lack of suficient and solid evidence-based medical support.Additionally,it was confirmed that a medical ethics review is not a prerequisite for the legality ofoff-label drug use.Finally, recommendations for off-label druguse in clinical practice were proposed,such as ensuring timeliness, adequacy,and formality of informed consent,adhering toa clear causal relationship construction,as wellas sorting out the scope and efectivenesslevel of evidence-based medicine to establish industry self-consistency.
Keywords: off-label drug use;medical damage liability; medical ethics review; empirical research
1 问题的提出
1.1相生相成的新药入市审批制度与超说明书用药
超说明书用药从某种程度上而言,它是药品监管与审批制度下必然的副产品。(剩余14818字)