甲状腺素替代治疗中、重度TBI后低T3综合征患者安全性分析

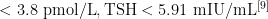
打开文本图片集
【中图分类号】R651【文献标志码】 B 【文章编号】1672-7770(2025)06-0673-05
Abstract:ObjectiveTo investigate the safety thyroid hormone replacement inpatientswith low T3 syndrome after traumatic brain injury(TBI).MethodsThe clinical data 7O6 patients with moderate orsevere TBI complicated with low T3syndrome whowere hospitalized in the fromJuly2018 January2024wereretrospectivelyanalyzed.Thepatients were dividedin studygroup and control groupacording whether they were supplementedwith exogenous thyroxine.There were 35O cases inthe control group and 356 cases inthe study group.The patients were followed up 6 months after discharge by telephoneand the adverse reactions occurred in the study group during medication. ResultsThere was no significant difference in baseline data between the two groups( P>0.05 ).Compared with the control group,the folow-updata 6months after discharge inthe study group were statisticallsignificantlydifferent from those in the control group( P<0.05 ).Univariate analysis adverse reactions in the study group showed that hisry diabetes,hisry hypertension,dosageanddays treatment wererisk facrs foradversereactions( P< 0.05).ConclusionsIn the clinical use thyroid hormone replacementtherapy forpatients with low T3 syndrome after moderateand severe TBI,individualizedand low-dosetreatment should be initiated,and serum thyroid function changes should be moniredregularly prevent drug-inducedadversereactionsduring theuse thyroid hormone replacement.
Key words:traumatic brain injury;low T3 syndrome;thyroid hormone;adverse reaction; safety
创伤性脑损伤(traumaticbraininjury,TBI)是指由外力引起的脑功能改变或其他的脑病理学改变,是世界范围内致死和致残的主要原因[1]。(剩余9243字)