阿替利珠单抗致上消化道出血1例

打开文本图片集
【中图分类号】R979.1 【文献标识码】A
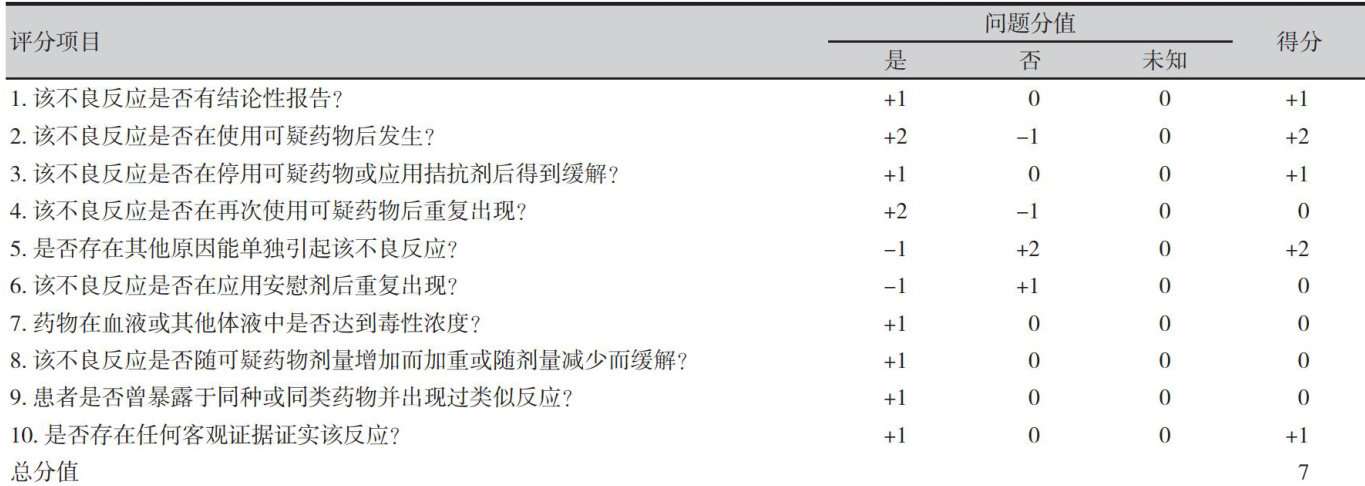
【Abstract】A 61-year-old female patient with advanced liver cancer, with a medical history of more than one year,had received various chemotherapy and targeted drugs in the past. Due to disease progression and adverse reactions, the treatment regimen was adjusted to atezolizumab monotherapy. After using atezolizumab for 16 cycles, the patient developed hematemesis with a hemoglobin level of 44g.L-1 . Gastrointestinal endoscopy revealed extensive erosion of the stomach and duodenum,and the patient was diagnosed with upper gastrointestinal bleeding. Considering the correlation with atezolizumab,a causality assessment using the Naranjo Assessment Scale was "likely". After drug discontinuation and symptomatic support treatment, thepatient's hemoglobin level recovered to 61g⋅L-1 . Thepatient was followed up at 8 weeks after discharge,and no digestive tract discomfort such as hematemesis and black stool occurred again.Atezolizumab has been on the market for a short time in China,and there is no related report of upper gastrointestinal bleeding with this drug in China. This case suggests that severe gastrointestinal adverse reactions of atezolizumab should be paid attention to in clinical treatment to ensure safe and effective medication for patients.
【Keywords】Atezolizumab; Upper gastrointestinal bleeding; Immune checkpoint inhibitors; Adverse drug reactions
阿替利珠单抗(atezolizumab)是一种程序性死亡配体1(programmed death-ligand1,PD-L1)免疫检查点抑制剂(immune checkpoint inhibitors,ICIs)。(剩余8971字)