乌司奴单抗相关不良反应文献病例分析

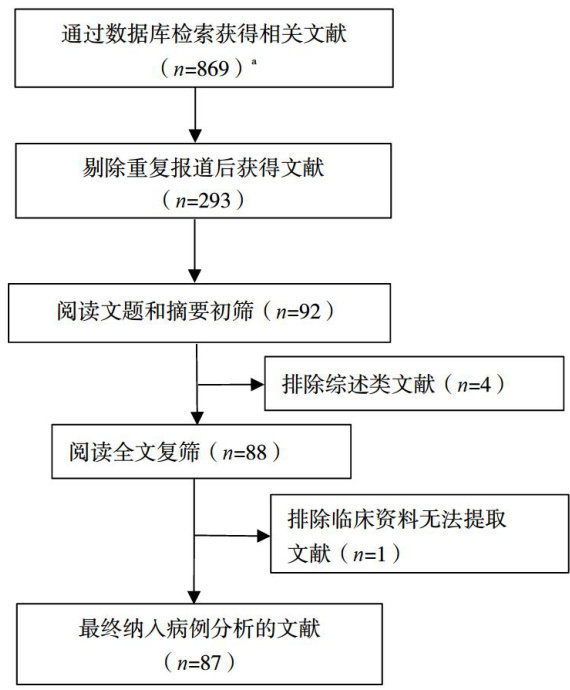
打开文本图片集
【中图分类号】R969.3 【文献标识码】A
【Abstract】 Objective To analyze the status and clinical characteristics of adverse drug reactions (ADRs) associated with ustekinumab and to provide a reference for clinical rational drug use.Methods PubMed,Web ofScience,Embase,CNKI, WanFang Data,and the VIPDatabase were searched for ADRs of ustekinumab,and the collected literature was further analyzed statistically, including the basic information of patients,the use of drugs,the induction time of adverse reactions, clinical manifestations,treatment and outcomes.Results A total of 87 articles were collected, reporting 115 ADRs,involving 106 patients,including 53 males and 53 females.The average age of the patients was (44.28±18.28) years old. The main underlying diseases were psoriasis (54 cases, 50.95% )and Crohn's disease (42 cases, 39.62% ).TheADRsmainly occurred within1 year (76 cases, 66.08% )aftertherapy.ADRsof ustekinumab were mainly skin and accessory (21.58%) )system injury,and followedbyrespiratory (16.11%) ,and nervous (13.07% ) system injury. A total of 100 patients recovered or improved after receiving dose reduction, drug withdrawal or symptomatic treatment; 3 patients did not improve,2 patients died,and 1 patient had no mention of improvement. Conclusion Clinicians and pharmacists should understand the characteristics of the ADRs of ustekinumab.More attention should be paid to ADRs of ustekinumab to ensure the safety of drug use.
【Keywords】Ustekinumab; Adverse drug reactions; Case report; Literature analysis
乌司奴单抗是一种人源化IgG1k单克隆抗体,作为白细胞介素(interleukin,IL)-12/23拮抗剂,可靶向结合IL-12和IL-23共有的 p40 亚基,阻断两者的生物学活性,进而抑制异常炎症反应[-2]。(剩余13381字)