基于FAERS数据库的药物相关血小板减少症信号挖掘

打开文本图片集
【中图分类号】R95 【文献标识码】A
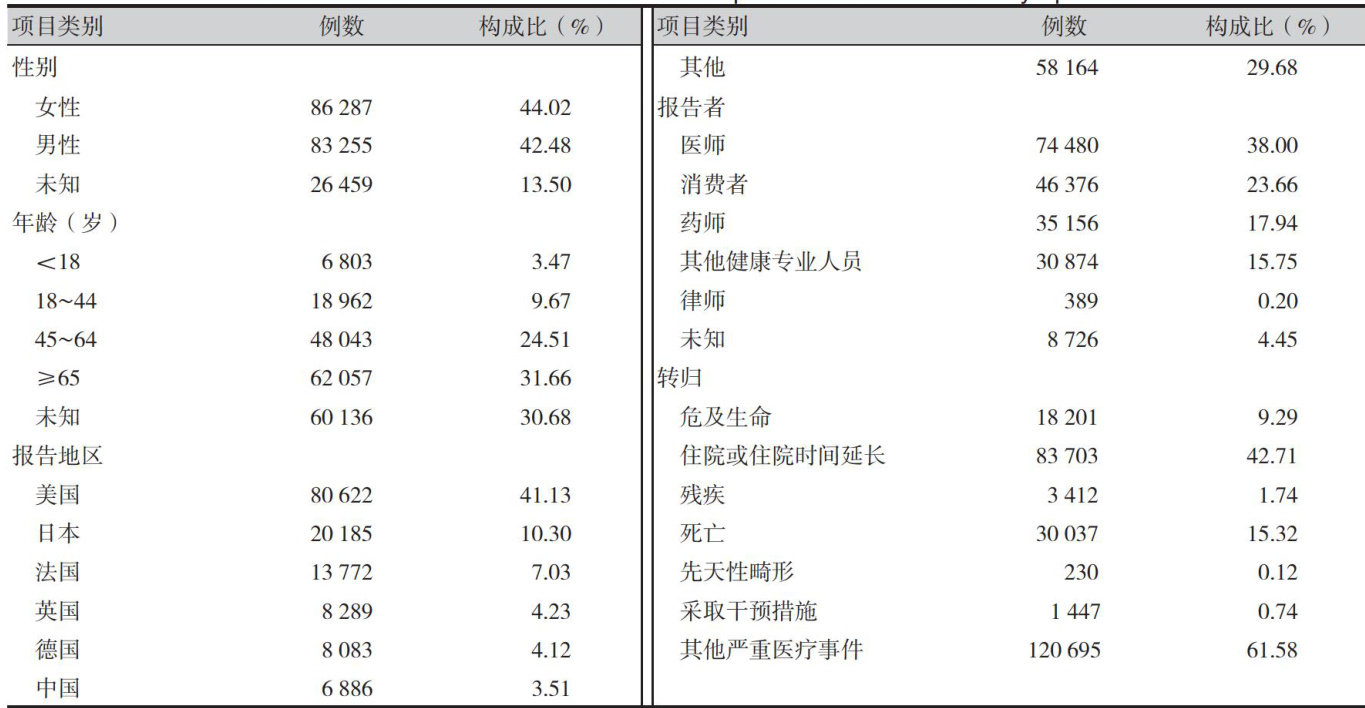
【Abstract】 Objective To identify drugs associated with thrombocytopenia based on the U.S. FDA Adverse Event Reporting System (FAERS) database,and to provide reference for safe clinical drug use.Methods Adverse drug event (ADE) reports related to thrombocytopenia were extracted from the FAERS database (Q1 2004 to Q3 2024).Signal detection and data analysis were performed using the reporting odds ratio (ROR) method, proportional reporting ratio (PRR) method,Bayesian confidence propagation neural network (BCPNN) method,and multi-item gamma Poissn shrinker (MGPS) method.The time-to-onset of ADEs were evaluated using median,interquartile range,and Weibull Distribution. Results A total of 199,497 thrombocytopeniarelated ADE reports were collcted,identifying 1,701 primarysuspected drugs,with 261 drugs showing positive signals.Excluding cases with missing gender data,female patients accounted for a slightly higher proportion (2 (44.02%) .Life-threatening or fatal outcomes were reported in 48,238 cases (24.61% ). The most frequently reported first-level Anatomical Therapeutic Chemical (ATC) groups were the drugs for the antineoplastic and immunomodulatingagentssystem (48.96%) ,followedbydrugsforbloodandhematopoieticorgans (7.70%) .The top three drugs byreport frequency were lenalidomide (12,165 cases), niraparib (4,996 cases),and ruxolitinib (4,978 cases). The three drugs with the highest ROR values were romiplostim [ ROR=28.22 -; 95%CI (27.20, 29.28)),eltrombopag 1ROR=25.24 , 95%CI (24.50,26.00)),and linezolid[ROR .=16.37 , 95%CI (15.65,17.13)). Lenalidomide was associated with the highest number of fatal outcomes (1,391 cases).Weibull shape parameter analysis indicated that most thrombocytopenia-inducing drugs exhibited an early failure pattern.Conclusion A total of 261 drugs associated with positive signals ofthrombocytopenia were monitored, with varying risk levels. Clinicians should closely monitor hematologic parameters during treatment, particularly for drugs that do not mention the risk of thrombocytopenia in their inserts,in order to prevent thrombocytopenia.
【Keywords】Thrombocytopenia; Adverse drug events; FAERS database; Pharmacovigilance; Signal mining
药物相关血小板减少症(drug-inducedthrombocytopenia,DITP)是指药物导致人体外周血液中血小板计数低于正常范围,并导致出血等症状的疾病。(剩余13813字)